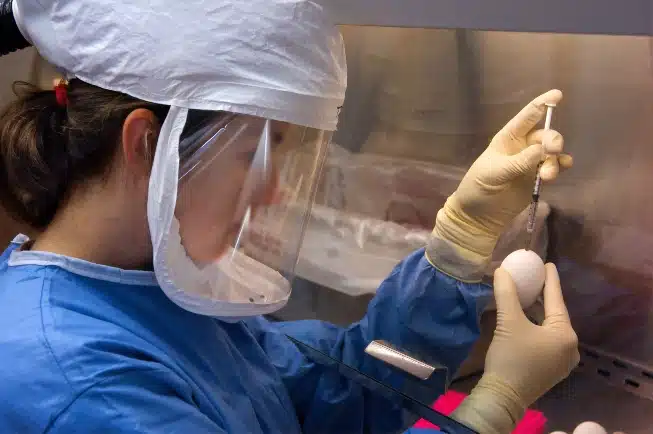
Plymouth Meeting-based Inovio has signed a manufacturing deal with Thermo Fisher for its experimental DNA-based Covid-19 vaccine candidate, writes John George for the Philadelphia Business Journal.
The biotechnology company is working on ensuring it can produce 100 million doses of INO-4800, its vaccine candidate, in 2021. The vaccine is still subject to approval by the Food and Drug Administration.
Inovio already has manufacturing deals in place with Germany-based Richter-Helm BioLogics and Ology Biosciences in Florida. It is also currently involved in litigation with Blue Bell-based VGXI, its previous contract manufacturer.
According to the new deal, Thermo Fisher will manufacture the INO-4800 drug substance and perform “fill and finish” of the product. The company projects it is capable of producing 100 million doses of the drug candidate annually at peak capacity.
“Inovio welcomes Thermo Fisher to our global consortium of commercial-scale vaccine manufacturers,” said Inovio CEO J. Joseph Kim. “Thermo Fisher’s global capabilities and scale will be central to our production progress (and) key to our ability to meet the urgent, global demand for a safe and effective vaccine against Covid-19.”
Inovio is now planning to begin late-stage testing of INO-4800 before the end of the month, pending FDA clearance.
Read more about Inovio at the Philadelphia Business Journal by clicking here.
You Might Also Like
Plymouth Meeting-Based Inovio Within Reach of COVID-19 Vaccine Breakthrough
Pandemic Could Mark Turning Point for Plymouth-based Inovio’s DNA Vaccine Technology
Positive Animal Study Data for Inovio’s COVID Vaccine Pushes Company’s Stock Up




















![ForAll_Digital-Ad_Dan_1940x300[59]](https://montco.today/wp-content/uploads/sites/2/2022/06/ForAll_Digital-Ad_Dan_1940x30059.jpg)




















![ForAll_Digital-Ad_Malaika_376x628[44]](https://montco.today/wp-content/uploads/sites/2/2022/06/ForAll_Digital-Ad_Malaika_376x62844.jpg)












































































